 The first energy level in which electrons orbit can hold up to two electrons. We know that oxygen has eight subatomic particles because its atomic number is eight. 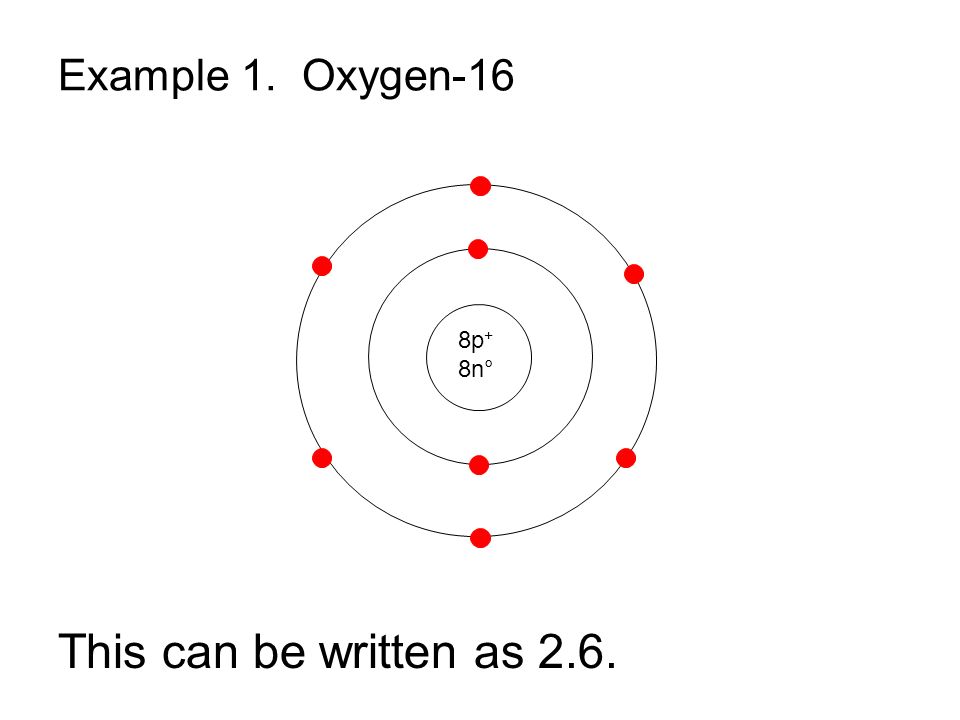
Bohr won a Nobel Prize in Physics for his contributions to our understanding of the structure of atoms and how. The Bohr Model has an atom consisting of a small, positively charged nucleus orbited by negatively charged electrons. About Transcript Calculating electron energy for levels n1 to 3. The Bohr model for oxygen shows eight protons and neutrons in the nucleus of the atom, with eight electrons orbiting the nucleus in two energy levels. the model laid the foundation for the quantum mechanical model of the atom. (b) If Thomson’s model of the atom were correct, the particles should have passed straight. (b) The energy of the orbit becomes increasingly less negative with increasing n. 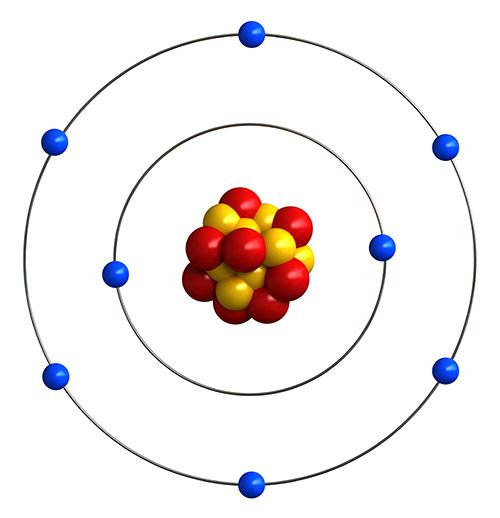
2 The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. The particles were produced by a sample of radium. In this state the radius of the orbit is also infinite. (a) A representation of the apparatus Rutherford used to detect deflections in a stream of particles aimed at a thin gold foil target. (Image credit: Pixabay) The Bohr model, introduced by Danish physicist Niels Bohr in. 
A model of an atom according to Niels Bohr. In this state the radius of the orbit is also infinite. 20.10 Occurrence, Preparation, and Properties of Sulfur. 7: A Summary of Rutherford’s Experiments. The Bohr model is neat, but imperfect, depiction of atom structure. The quantity in parentheses is a unit of energy known as a Rydberg, which \(\approx 13.6eV\).īohr reasoned that the hydrogen atom doesn't radiate away all of its energy, because the lowest energy level (which we would now call the ground state) corresponds to one wavelength fitting in the orbit, so \(n=1\) is the lowest it can go. 20.9 Occurrence, Preparation, and Compounds of Oxygen. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
